The unprecedented health crisis caused by the coronavirus disease 2019 (COVID-19) pandemic has affected over 247 million individuals to date. Monoclonal antibodies (mAbs) that target the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike glycoprotein have been found to have good neutralization efficacy in the treatment of Ebola virus, SARS, middle east respiratory syndrome (MERS), infections in the past as well as in treating COVID-19 as indicated by recent studies.
.jpg)
The viral spike protein has been the key target for the development of therapeutic mAbs. Most neutralizing antibodies (NAbs) bind directly to the spike protein's receptor-binding domain (RBD), although some NAbs bind to the N-terminal domain. Various sources from which NAbs are derived include SARS patients, memory B cells from SARS-CoV-2 convalescent patients, immunized humanized mice encoding the human immunoglobulin repertoire, single domain human antibodies from existing libraries, alpaca nanobodies, and phage display antibody libraries.
SARS-CoV-2 NAbs from a pre-pandemic human combinatorial antibody library
In an article published in the open-access journal Advance Engineering, a multinational team of researchers reported the selection and characterization of 3 potent SARS-CoV-2 antibodies – S-E6, S-B8, and S-D4 – from a pre-COVID-19 human combinatorial antibody library that was established 20 years ago. The antibodies bind to the RBD of the spike protein, thus competing with the human angiotensin-converting enzyme 2 (hACE2) receptor. Compared to antibodies derived from COVID-19 patients with low somatic hypermutation (SHM), these 3 antibodies had more than 13–22 SHMs. Many of these mutations were involved in specific interactions with SARS-CoV-2 spike RBD.
"The combinatorial antibody library technology allows the same evolutionary process to be performed in vitro as it restores the "fossil record" of an individual's antibody response in a test tube."
The findings of this study show that a highly diverse combinatorial antibody library can mimic the natural immunity selection process and allow the detection of unexpected, high-affinity antibodies targeting the spike protein. It also permits the selection of binding molecules with chemistries not accessible during in vivo selection.
Presence of SARS-CoV-2 NAbs in a pre-pandemic library could indicate past epidemics involving similar coronaviruses
Combinatorial antibody libraries can immensely simplify the antibody discovery process and also enable the documentation of the antibody response history of an individual. In addition, the COVID-19 pandemic has prompted scientists across the globe to use this technology more widely to mitigate the unprecedented public health crisis related to the pandemic.
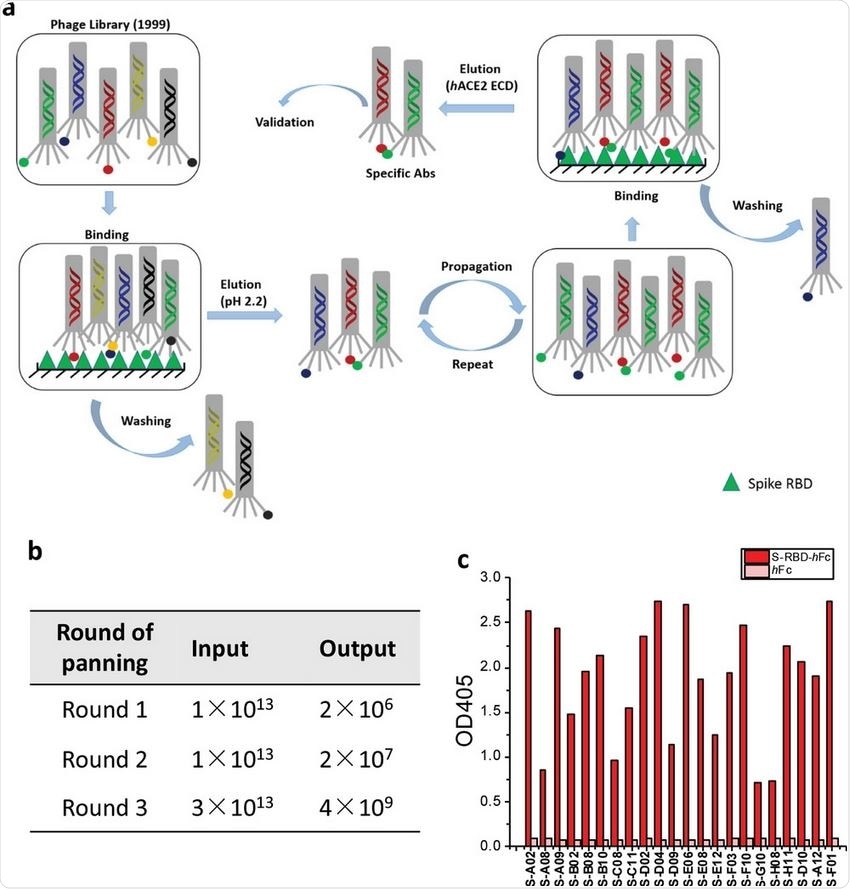
In this study, a combinatorial human antibody library constructed in 1999 was used to discover three potent antibodies that can selectively bind to the spike protein of SARS-CoV-2 and neutralize the virus. When combined with antibodies taken from convalescent patients or convalescent plasma, these antibodies derived from combinatorial libraries proved to be an effective therapeutic option and a possible antidote to immune escape by newly emerging variants of SARS-CoV-2.
"Mixtures of antibodies targeting distinct epitopes can be used to overcome such immune escape.Since a complete antibody repertoire of an individual is the starting reservoir of all possibilities, one can also learn much more about the origins and evolution of an immune response against viral challenge when it is studied."
The presence of highly potent NAbs in a pre-pandemic library of "healthy" donors could indicate prior exposure of donors to similar coronaviruses in the past or to an antigenic surface that closely resembles the SARS-CoV-2 RBD. According to the authors, it still remains to be seen if these antibodies result from background immunity. Since there is a random coupling of VH and VL sequences in the original combinatorial antibody library, the isolated antibodies do not fully represent the natural selection process of the human B cell repertoire.
Identifying these antibodies with high SHM in a pre-COVID library raises interesting questions about their origin and evolution and their reactivity to SARS-CoV-2. In addition, a better understanding of immune response regarding interactions between NAbs and their SARS-CoV-2 binding epitopes can provide a blueprint for the design of next-generation vaccines.
"We cannot rule out potential implications of such findings from libraries made decades ago concerning the origin of the viruses currently circulating."
- Qiang, M., Ma, P., Li, Y., Liu, H., Harding, A., Min, C., Wang, F., Liu, L., Yuan, M., Ji, Q., Tao, P., Shi, X., Li, Z., Li, T., Wang, X., Zhang, Y., Wu, N. C., Lee, C.-C. D., Zhu, X., Gilbert-Jaramillo, J., Zhang, C., Saxena, A., Huang, X., Wang, H., James, W., Dwek, R. A., Wilson, I. A., Yang, G., Lerner, R. A., Neutralizing Antibodies to SARS-CoV-2 Selected from a Human Antibody Library Constructed Decades Ago. Adv. Sci. 2021, 2102181. https://doi.org/10.1002/advs.202102181, https://onlinelibrary.wiley.com/doi/10.1002/advs.202102181
Posted in: Medical Science News | Medical Research News | Disease/Infection News
Tags: Angiotensin, Angiotensin-Converting Enzyme 2, Antibodies, Antibody, Antibody Discovery, Antidote, Antigen, B Cell, Cell, Convalescent Plasma, Coronavirus, Coronavirus Disease COVID-19, Efficacy, Enzyme, Evolution, Glycoprotein, Immune Response, immunity, Immunoglobulin, in vitro, in vivo, Nanobodies, Pandemic, Protein, Public Health, Receptor, Respiratory, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Spike Protein, Syndrome, Virus

Written by
Susha Cheriyedath
Susha has a Bachelor of Science (B.Sc.) degree in Chemistry and Master of Science (M.Sc) degree in Biochemistry from the University of Calicut, India. She always had a keen interest in medical and health science. As part of her masters degree, she specialized in Biochemistry, with an emphasis on Microbiology, Physiology, Biotechnology, and Nutrition. In her spare time, she loves to cook up a storm in the kitchen with her super-messy baking experiments.
Source: Read Full Article



