The ongoing coronavirus disease 2019 (COVID-19) pandemic is caused by the rapid spread of the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). SARS-CoV-2, which was first reported in Wuhan, China in 2019, is a positive-sense ribonucleic acid (RNA) virus belonging to the family Coronaviridae.
 Study: Secreted SARS-CoV-2 ORF8 modulates the cytokine expression profile of human macrophages. Image Credit: Yarchanka Siarhei / Shutterstock.com
Study: Secreted SARS-CoV-2 ORF8 modulates the cytokine expression profile of human macrophages. Image Credit: Yarchanka Siarhei / Shutterstock.com
ORFs of the SARS-CoV-2 genome
Scientists have explained that the RNA genome of SARS-CoV-2 is approximately 30 kilobases (kb) in size and contains 12 major ORFs. These ORFs are associated with polyprotein processing and the translation events that encode upwards regulation of 30 proteins.
ORFs are classified as replication proteins, structural proteins, and accessory proteins based on their function. The products of Orf1a/b polyprotein are involved with the replication process. The structural proteins of SARS-CoV-2, of which include the spike (S), envelope (E), membrane (M), and nucleocapsid (N) proteins, are the most important components of this virus. The expression of ORF3a, ORF6, ORF7a/b, ORF8, ORF9b/c, as well as the ORF10 region result in the production of accessory proteins.
Previous studies have reported that the accessory proteins of coronaviruses are not always essential for viral replication and that the encoding regions associated with the expression of accessory proteins are less likely to remain conserved among different viral species. Despite these observations, these proteins play an important role in immune modulation. Other important functions of these proteins are associated with virulence, pathogenesis, and host adaptation.
Role of ORFs in SARS-CoV-2 infection
Scientists have found that during the initial phase of the COVID-19 pandemic, the SARS-CoV-2 ORF8 gene was an early hotspot of mutational change. Similar to that of SARS-CoV-1, various studies have shown that large gene deletions in the ORF8 of SARS-CoV-2, as well as L84S polymorphisms, are related to changes in disease severity.
It is extremely important to understand the functions of the SARS-CoV-2 ORF8 polypeptide, as it would provide better insights into viral adaptation and pathogenesis. Previous studies have shown that even though a complete ORF8 gene was not necessary for SARS-CoV-1 replication, intact ORF8 polypeptide was found to be glycosylated and involved in the blocking of innate immune signaling. This gene is found in the endoplasmic reticulum (ER).
Bioinformatics analyses indicate the presence of cleavable signal peptide (SP) in the ORG8 of SARS-CoV-2. This result was supported by another group of researchers who observed SP-dependent secretion of ORF8 in both transfected cell culture supernatants and the serum of COVID-19 patients.
Earlier studies also indicate that this ORF8 inhibits various stages of the interferon (IFN) signaling pathway. Further, the ORF8 gene is also reported to be involved with immune evasion via direct interaction with and down-regulating major histocompatibility complex (MHC-)I molecules in various cell types. The ORF8 of SARS-CoV-2 has also been linked with the cytokine storms associated with COVID-19 as a result of its interaction with the interleukin (IL)-17 receptor.
About the study
A new study available on the preprint server bioRxiv* determined the production of the SARS-CoV-2 SP-dependent ORF8.
This study also revealed that the secreted ORF8, which is primarily a disulfide-linked dimer that is glycosylated at position 78, is an immunomodulator. In other words, ORF8 can modulate the cytokine response of human blood monocyte-derived macrophages (MDMs) in a manner that depends on ORF8 L84S sequence polymorphisms.
In agreement with previous reports, the authors of the current study have also reported SP-dependent secretion of wild type ORF8 FLAG construct (WT-f) and L84S ORF8. They have also successfully identified a biosynthetic pathway connected to mature glycosylation and secretion of a disulfide-linked dimer.
The researchers have also detected smaller species, which are about 11 kilodaltons (kDa) in size and of an unknown origin, that retained the C-terminal FLAG tag, thus indicating its association with an N-terminal cleavage event.
The present study revealed that an over-expression of ORF8 induces ER stress. It also showed that no major positive or negative effect on IFN secretion, response, or NF-κB signaling in A549, X293T, and THP-1 cells respectively occurred as a result of OFR8 secretion.
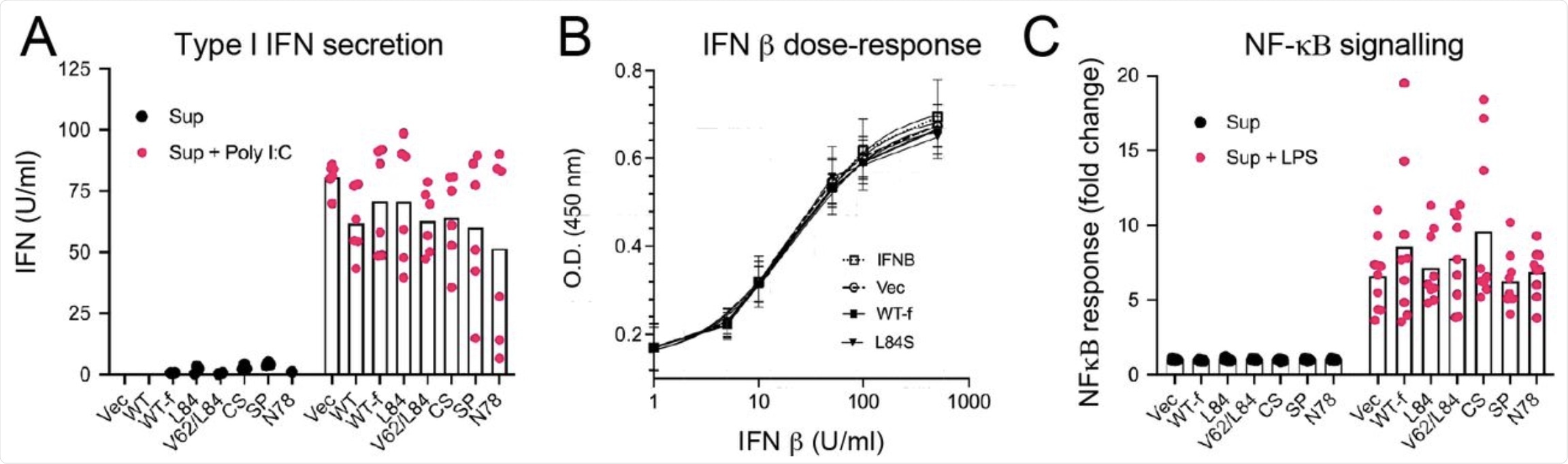 Secreted ORF8 does not affect NF-κB or IFN signalling. (A) ORF8 containing supernatants were added onto A549 cells, either alone or in combination with Poly I:C (2.5 μg/ml). 24 h later supernatants from A549s were harvested and assayed for type I Interferon using HEK-Blue IFNα/β cells. Type I IFN amounts were extrapolated from a standard curve and values below baseline are not plotted. Data points are from two independent experiments each done in triplicate. Bar indicates the mean. (B) An IFN β dose-response curve analysis was performed on HEK -Blue IFN α/β cells in the absence (IFN) or presence of control (Vec) or ORF8 (WT-f or L84S)-containing supernatants. Data are the mean ± SD of three independent experiments each performed in triplicate and have been fitted with a three-parameter agonist-response curve in Graphpad Prism. All R2 values were > 0.93 and the estimated EC50 values were not significantly different (p > 0.05). (C) Control or various ORF8-containing supernatants were added to THP-1 Blue cells in the presence or absence of LPS (10 ng/ml) and NF-κB response was measured using QuantiBlue. Data points from three independent experiments done in triplicate. Bars indicate the mean.
Secreted ORF8 does not affect NF-κB or IFN signalling. (A) ORF8 containing supernatants were added onto A549 cells, either alone or in combination with Poly I:C (2.5 μg/ml). 24 h later supernatants from A549s were harvested and assayed for type I Interferon using HEK-Blue IFNα/β cells. Type I IFN amounts were extrapolated from a standard curve and values below baseline are not plotted. Data points are from two independent experiments each done in triplicate. Bar indicates the mean. (B) An IFN β dose-response curve analysis was performed on HEK -Blue IFN α/β cells in the absence (IFN) or presence of control (Vec) or ORF8 (WT-f or L84S)-containing supernatants. Data are the mean ± SD of three independent experiments each performed in triplicate and have been fitted with a three-parameter agonist-response curve in Graphpad Prism. All R2 values were > 0.93 and the estimated EC50 values were not significantly different (p > 0.05). (C) Control or various ORF8-containing supernatants were added to THP-1 Blue cells in the presence or absence of LPS (10 ng/ml) and NF-κB response was measured using QuantiBlue. Data points from three independent experiments done in triplicate. Bars indicate the mean.
The researchers also observed that the cell type has a significant effect on the secreted ORF8 in modulating IL-6 production by human primary MDMs. However, this observation was not re-evaluated using A549 cells, which can produce IL-6.
The authors of the current study showed that secreted ORF8 can modulate cytokine secretion by MDMs, which possess immunomodulatory “virokine” function. They reported that ORF8 secretion altered cytokine expression from primary CSF1-derived human macrophages, by decreasing IL-6 and IL-8 secretion.
The results of this study also suggest that two common ORF8 polymorphisms in the early phase of the pandemic had different effects on the L84S variant that reduced the clinical severity of the disease.
Conclusions and future research
The mechanism by which ORF8 alters cytokine expression has yet to be determined. Since the present study did not evaluate purified ORF8 polypeptides, the possibility of ORF8 synthesis in cells stimulating the release of factors that could modify MDM function could not be excluded.
In conclusion, ORF8 sequence polymorphisms appear to significantly affect the virulence of SARS-CoV-2; therefore, these genetic changes should be monitored through sequencing-based surveillance.
*Important notice
bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
- Kriplani, N., Clohisey, S., Fonseca, S., et al. (2021) Secreted SARS-CoV-2 ORF8 modulates the cytokine expression profile of human macrophages. bioRxiv. doi:10.1101/2021.08.13.456266. https://www.biorxiv.org/content/10.1101/2021.08.13.456266v2
Posted in: Medical Science News | Medical Research News | Medical Condition News | Disease/Infection News | Healthcare News
Tags: Bioinformatics, Blood, Cell, Cell Culture, Coronavirus, Coronavirus Disease COVID-19, Cytokine, Gene, Genetic, Genome, Glycosylation, Immunomodulatory, Membrane, Monocyte, Pandemic, Receptor, Research, Respiratory, Ribonucleic Acid, RNA, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Signaling Pathway, Stress, Syndrome, Translation, Virus

Written by
Dr. Priyom Bose
Priyom holds a Ph.D. in Plant Biology and Biotechnology from the University of Madras, India. She is an active researcher and an experienced science writer. Priyom has also co-authored several original research articles that have been published in reputed peer-reviewed journals. She is also an avid reader and an amateur photographer.
Source: Read Full Article



