Since the onset of the CRISPR genetic editing revolution, scientists have been working to leverage the technology in the development of gene drives that target pathogen-spreading mosquitoes such as Anopheles and Aedes species, which spread malaria, dengue and other life-threatening diseases.
Much less genetic engineering has been devoted to Culex genus mosquitoes, which spread devastating afflictions stemming from West Nile virus—the leading cause of mosquito-borne disease in the continental United States—as well as other viruses such as the Japanese encephalitis virus (JEV) and the pathogen causing avian malaria, a threat to Hawaiian birds.
University of California San Diego scientists have now developed several genetic editing tools that help pave the way to an eventual gene drive designed to stop Culex mosquitoes from spreading disease. Gene drives are designed to spread modified genes, in this case those that disable the ability to transmit pathogens, throughout the targeted wild population.
As detailed in the journal Nature Communications, Xuechun Feng, Valentino Gantz and their colleagues at Harvard Medical School and National Emerging Infectious Diseases Laboratories developed a Cas9/guide-RNA expression “toolkit” designed for Culex mosquitoes. Since such little attention in genetic engineering has been devoted to Culex mosquitoes, the researchers were required to develop their toolkit from scratch, starting with a careful examination of the Culex genome.
https://youtube.com/watch?v=5jezBBdL050%3Fcolor%3Dwhite
“My coauthors and I believe that our work will be impactful for scientists working on the biology of the Culex disease vector since new genetic tools are deeply needed in this field,” said Gantz, an assistant research scientist in the Division of Biological Sciences at UC San Diego. “We also believe the scientific community beyond the gene drive field will welcome these findings since they could be of broad interest.”
While Culex mosquitoes are less problematic in the United States, they are much more of a health risk in Africa and Asia, where they transmit the worm causing filariasis, a disease that can lead to a chronic debilitating condition known as elephantiasis.
The researchers also demonstrated that their tools could work in other insects.
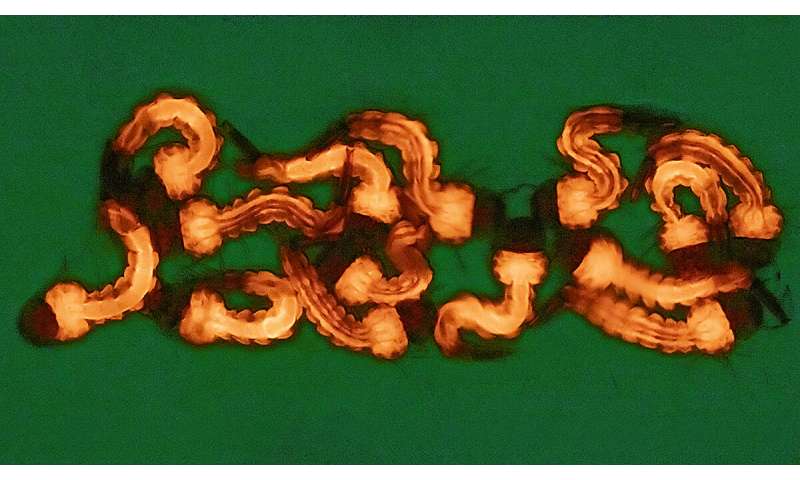
“These modified gRNAs can increase gene drive performance in the fruit fly and could potentially offer better alternatives for future gene drive and gene-editing products in other species,” said Gantz.
Source: Read Full Article